Bulletin of Botanical Research ›› 2026, Vol. 46 ›› Issue (2): 259-269.doi: 10.7525/j.issn.1673-5102.2026.02.006
• Original Paper • Previous Articles Next Articles
Haiyan CAO1, Kaiwen TIAN2, Xiaoyu JIA1, Xuefeng HAO1, Zhuping JIN2( )
)
Received:2026-02-18
Online:2026-03-20
Published:2026-04-02
Contact:
Zhuping JIN
E-mail:jinzhuping@sxu.edu.cn
CLC Number:
Haiyan CAO, Kaiwen TIAN, Xiaoyu JIA, Xuefeng HAO, Zhuping JIN. The Role of AtMST1 in Regulating Salt Tolerance via H2S Synthesis in Arabidopsis Revealed by CRISPR/Cas9 Knockout[J]. Bulletin of Botanical Research, 2026, 46(2): 259-269.
Add to citation manager EndNote|Ris|BibTeX
URL: https://bbr.nefu.edu.cn/EN/10.7525/j.issn.1673-5102.2026.02.006
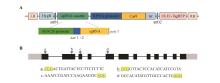
Fig.1
Schematic diagram of pKI1.1 R vector and target sites of AtMST1 geneA. Structure of pKI1.1 R vector; LB. T-DNA left border; RB. T-DNA right border; HygR. Hygromycin resistance gene; U6.26 promoter; U6.26 promoter driving sgRNA expression. B. Gene structure of AtMST1 and sgRNA target sites; gray boxes. 5' and 3'-untranslated regions; black boxes. Exons; black lines. Introns; lowercase letters. Locations of gRNA target sequences.
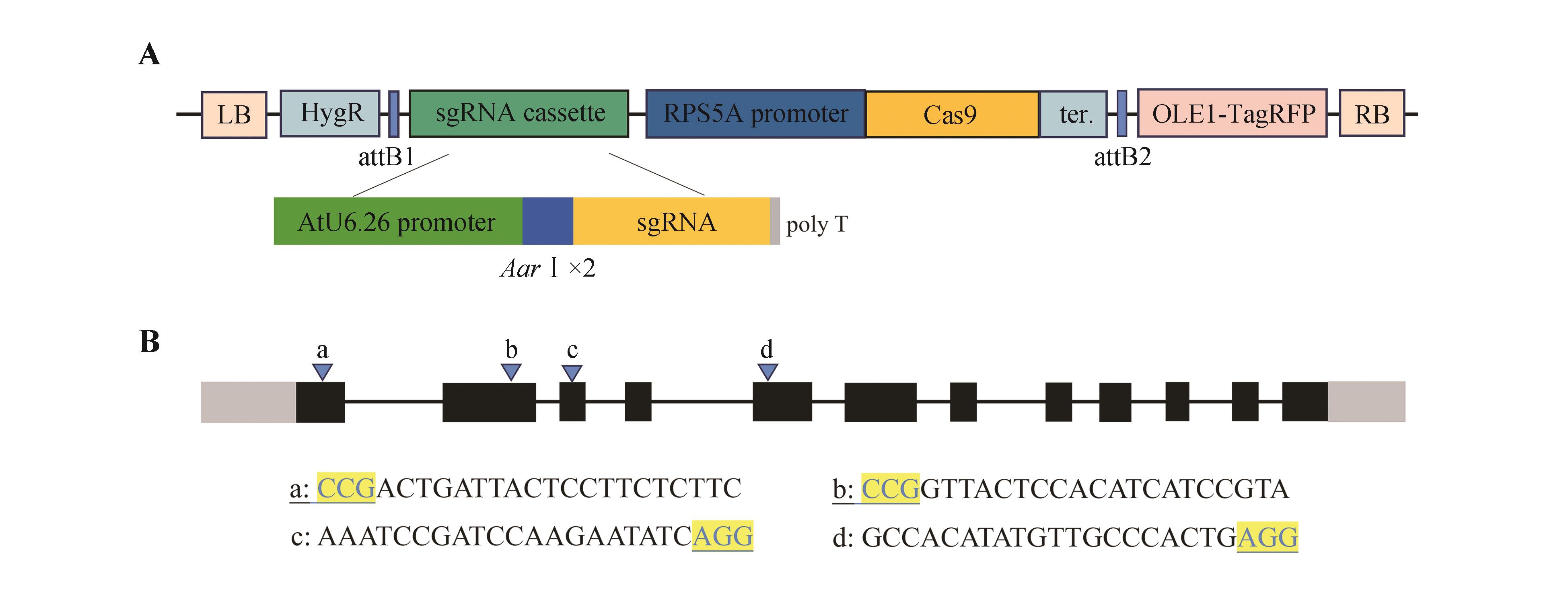
Table 1
All primers appearing in the text
引物名称 Primer name | 引物编号 Primer ID | 引物序列5′→3′ Primer sequence | 用途 Purpose |
|---|---|---|---|
| AtMST1-gRNA1-F1 | pp2043 | ATTGGAAGAGAAGGAGTAATCAGT | gRNA1靶点扩增 |
| AtMST1-gRNA1-R1 | pp2044 | AAACACTGATTACTCCTTCTCTTC | |
| AtMST1-gRNA2-F2 | pp2045 | ATTGGTTACTCCACATCATCCGTA | gRNA2靶点扩增 |
| AtMST1-gRNA2-R2 | pp2046 | AAACTACGGATGATGTGGAGTAAC | |
| AtMST1-gRNA3-F3 | pp2047 | ATTGAAATCCGATCCAAGAATATC | gRNA3靶点扩增 |
| AtMST1-gRNA3-R3 | pp2048 | AAACGATATTCTTGGATCGGATTT | |
| AtMST1-gRNA4-F4 | pp2049 | ATTGGCCACATATGTTGCCCACTG | gRNA4靶点扩增 |
| AtMST1-gRNA4-R4 | pp2050 | AAACCAGTGGGCAACATATGTGGC | |
| AtMST1-F | pp2247 | ATGGCCTCGACCCTTTTCTC | AtMST1阳性植株编辑位点鉴定 |
| AtMST1-R | pp2250 | GACCTCCATCGAGCACCCA | |
| CAS9-F | pp2060 | GCCTGTTCGGAAACCTGAT | Cas9鉴定 |
| CAS9-R | pp2061 | GTAGCCGTTCTTGCTCTGG | |
| pKI1.1R-F | pp1729 | TGGGAAAGAACAATAGTAT | 载体引物,阳性克隆的鉴定 |
Fig.4
Molecular identification of AtMST1-CRISPR gene-edited plantsM. 2 000 DNA marker; CK. Negative control; WT. Wild type; numbers. Representative independent transgenic lines; CR. CRISPR; CR1,CR2,CR3,and CR4. Four independent gene-editing vectors. Upper panel(A-D). PCR products amplified with primers CAS9-F/CAS9-R, showing the presence of the Cas9 gene; lower panel(E-H). PCR products amplified with primers pKI1.1R-F/AtMST1-R,confirming the insertion of the target gene fragment.
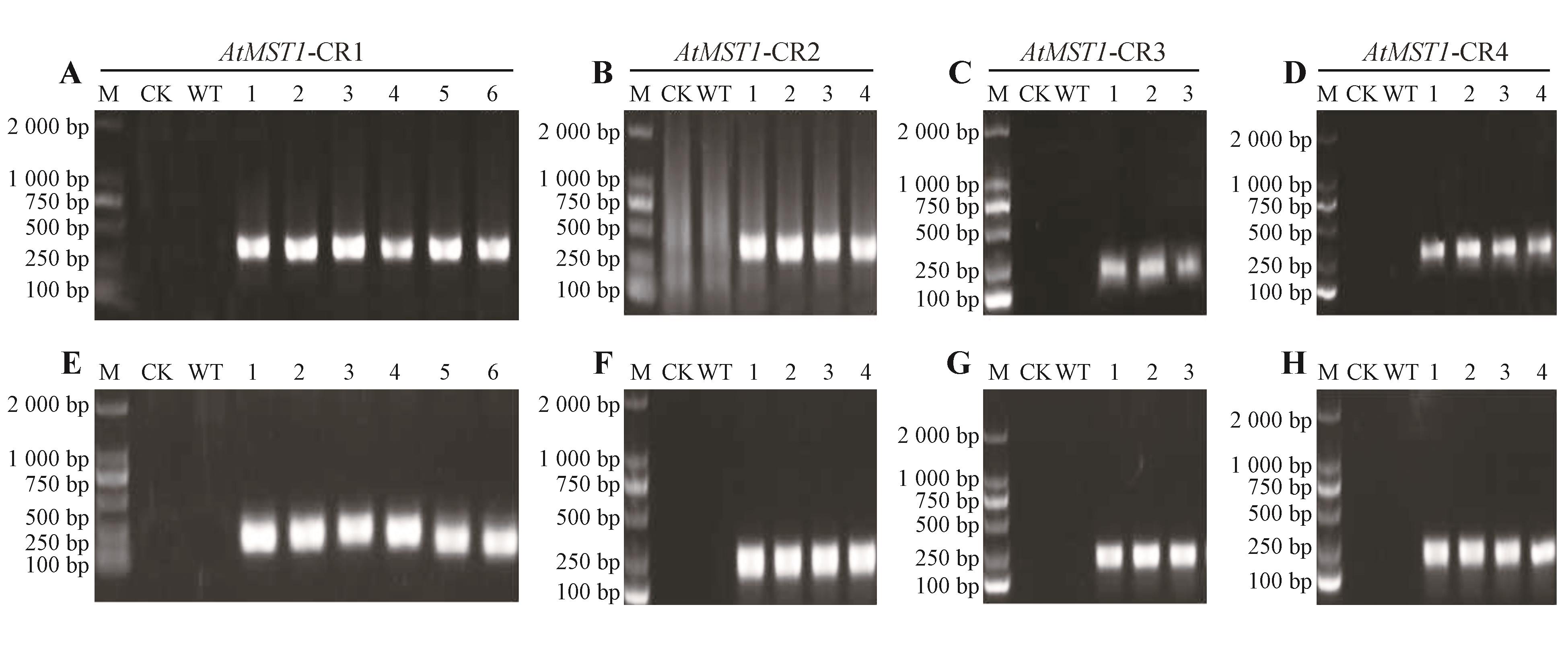
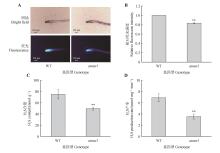
Fig.6
The H2S production rate and content in atmst1 mutantsA. Representative images of H2S detection in roots using the 7-Azido-4-methylcoumarin(AzMc) fluorescent probe;B.Quantitative analysis of fluorescence intensity shown in(A) using Image J software;C. H2S content in wild-type(WT) and atmst1 mutant plants measured by the methylene blue assay;D.Endogenous H2S production rate in WT and atmst1 mutants determined by the methylene blue method. Data were presented as mean±SD. Asterisks indicated significant differences(**.P<0.01) as determined by Student’s t-test.
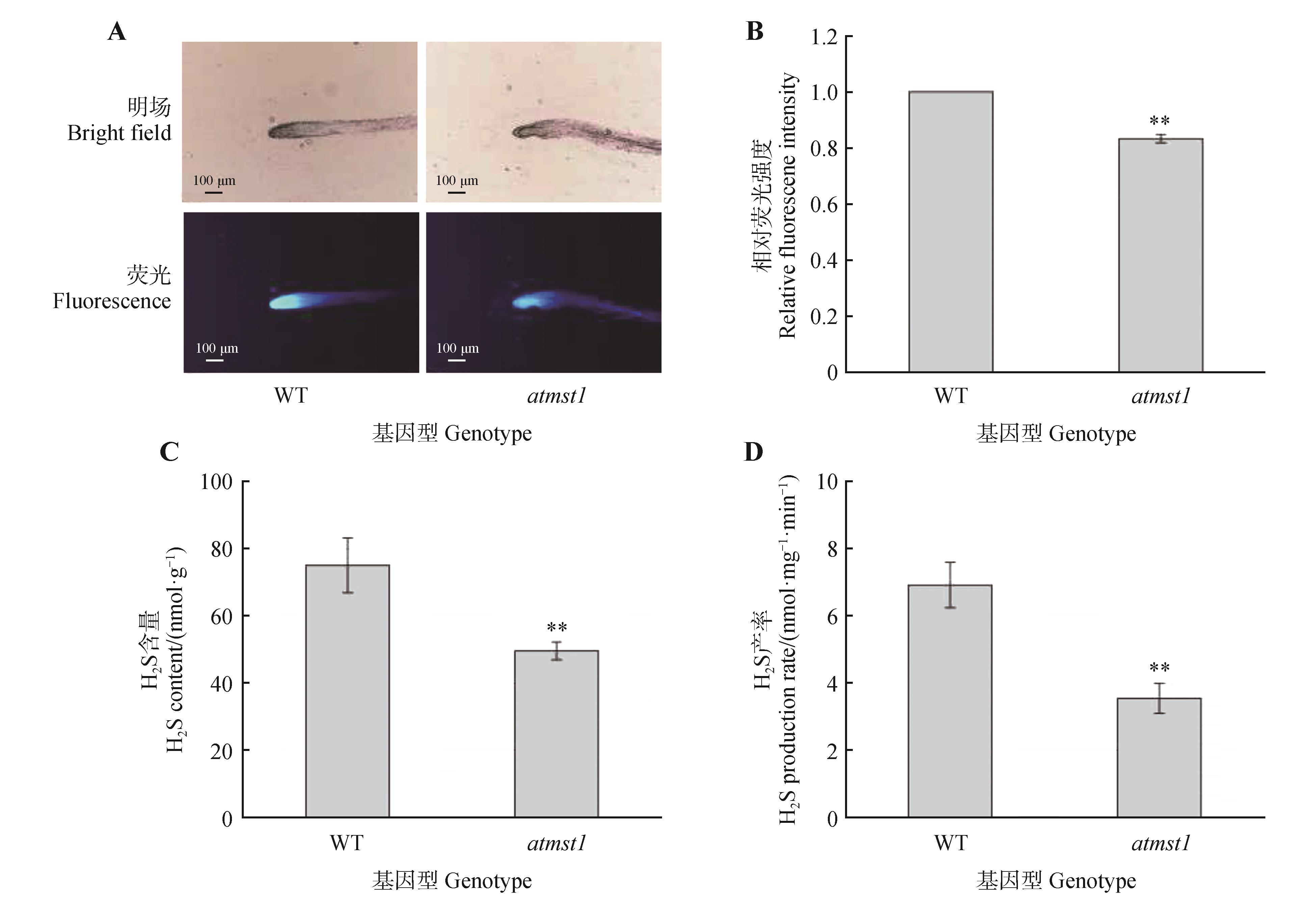
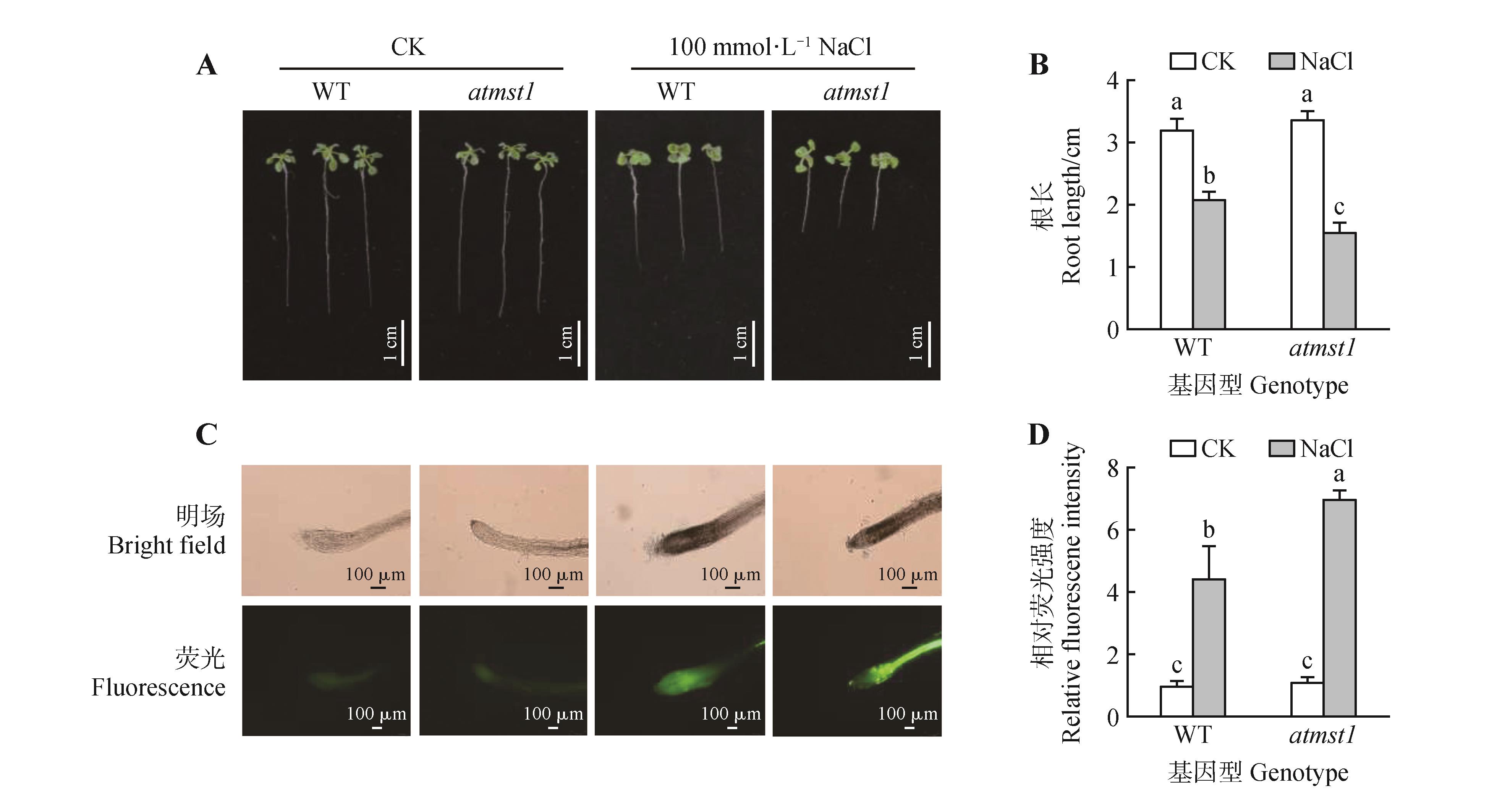
Fig.7
Loss of AtMST1 function led to salt sensitivity and increased ROS accumulation in Arabidopsis seedlingsA. Phenotypes of WT and atmst1 mutant plants grown under control(CK) or 100 mmol?L-1 NaCl conditions for 14 days; B. Quantitative analysis of root length for seedlings shown in(A); C. Representative fluorescence microscope images of seedling root tips stained with the ROS fluorescent probe DCFH-DA; D. Quantitative analysis of relative fluorescence intensity of ROS in root tips of WT and atmst1 seedlings under salt stress. Data were mean±SD, different lowercase letters indicated significant differences(P<0.05).
| [1] | WANG R.Physiological implications of hydrogen sulfide:a whiff exploration that blossomed[J].Physiological Reviews,2012,92(2):791-896. |
| [2] | ZHAO Y J, LI T, JIANG Z G,et al.The miR-9-5p/CXCL11 pathway is a key target of hydrogen sulfide-mediated inhibition of neuroinflammation in hypoxic ischemic brain injury[J].Neural Regeneration Research,2024,19(5):1084-1091. |
| [3] | DONG X Z, ZHANG H, DUAN P,et al.An injectable and adaptable hydrogen sulfide delivery system for modulating neuroregenerative microenvironment[J].Science Advances,2023,9(51):eadi1078. |
| [4] | CIRINO G, SZABO C, Papapetropoulos A.Physiological roles of hydrogen sulfide in mammalian cells,tissues,and organs[J].Physiological Reviews,2023,103(1):31-276. |
| [5] | SUN H J, LU Q B, ZHU X X,et al.Pharmacology of hydrogen sulfide and its donors in cardiometabolic diseases[J].Pharmacological Reviews,2024,76(5):846-895. |
| [6] | ZHAO Y Q, HU K D, YAO G F,et al.A D-cysteine desulfhydrase,SlDCD2,participates in tomato fruit ripening by modulating ROS homoeostasis and ethylene biosynthesis[J].Horticulture Research,2023,10(3):uhad014. |
| [7] | XIANG Z X, LI W, LU Y T,et al.Hydrogen sulfide alleviates osmotic stress-induced root growth inhibition by promoting auxin homeostasis[J].The Plant Journal,2023,114(6):1369-1384. |
| [8] | ZHOU M J, ZHANG J, SHEN J,et al.Hydrogen sulfide-linked persulfidation of ABI4 controls ABA responses through the transactivation of MAPKKK18 in Arabidopsis [J].Molecular Plant,2021,14(6):921-936. |
| [9] | LISJAK M, SRIVASTAVA N, TEKLIC T,et al.A novel hydrogen sulfide donor causes stomatal opening and reduces nitric oxide accumulation[J].Plant Physiology and Biochemistry,2010,48(12):931-935. |
| [10] | CHEN J, WU F H, WANG W H,et al.Hydrogen sulphide enhances photosynthesis through promoting chloroplast biogenesis,photosynthetic enzyme expression,and thiol redox modification in Spinacia oleracea seedlings[J].Journal of Experimental Botany,2011,62(13):4481-4493. |
| [11] | HAO X F, JIN Z P, WANG Z Q,et al.Hydrogen sulfide mediates DNA methylation to enhance osmotic stress tolerance in Setaria italica L.[J].Plant and Soil,2020,453:355-370. |
| [12] | AROCA A, ZHANG J, XIE Y J,et al.Hydrogen sulfide signaling in plant adaptations to adverse conditions:molecular mechanisms[J].Journal of Experimental Botany,2021,72(16):5893-5904. |
| [13] | CAO H Y, LIANG Y L, ZHANG L P,et al.AtPRMT5-mediated AtLCDmethylation improves Cd2+ tolerance via increased H2S production in Arabidopsis [J].Plant Physiology,2022,190(4):2637-2650. |
| [14] | 裴雁曦.植物中的气体信号分子硫化氢:无香而立,其臭如兰[J].中国生物化学与分子生物学报,2016,32(7):721-733. |
| PEI Y X.Gasotransmitter hydrogen sulfide in plant:stinking to high heaven,but refreshing to fine life[J].Chinese Journal of Biochemistry and Molecular Biology,2016,32(7):721-733. | |
| [15] | JARABAK R, WESTLEY J.Steady-state kinetics of 3-mercaptopyruvate sulfurtransferase from bovine kidney[J].Archives of Biochemistry and Biophysics,1978,185(2):458-465. |
| [16] | BORDO D, BORK P.The rhodanese/Cdc25 phosphatase superfamily:sequence-structure-function relations[J].The EMBO Reports,2002,3(8):741-746. |
| [17] | RAN M X, LI Q B, XIN Y F,et al.Rhodaneses minimize the accumulation of cellular sulfane sulfur to avoid disulfide stress during sulfide oxidation in bacteria[J].Redox Biology,2022,53:102345. |
| [18] | PAPENBROCK J, SCHMIDT A.Characterization of a sulfurtransferase from Arabidopsis thaliana [J].European Journal of Biochemistry,2000,267(1):145-154. |
| [19] | CROPPI G, ZHOU Y Y, YANG R,et al.Discovery of an inhibitor for bacterial 3-mercaptopyruvate sulfurtransferase that synergistically controls bacterial survival[J].Cell Chemical Biology,2020,27(12):1483-1499. |
| [20] | BAUER M, DIETRICH C, NOWAK K,et al.Intracellular localization of Arabidopsis sulfurtransferases[J].Plant Physiology,2004,135(2):916-926. |
| [21] | BARTELS A, MOCK H P, PAPENBROCK J.Differential expression of Arabidopsis sulfurtransferases under various growth conditions[J].Plant Physiology and Biochemistry,2007,45(3/4):178-187. |
| [22] | GURETZKI S, PAPENBROCK J.Characterization of the sulfurtransferase family from Oryza sativa L[J].Plant Physiology and Biochemistry,2011,49(9):1064-1070. |
| [23] | HAO X F, CAO H Y, XIE M J,et al.AtMST1:a novel hydrogen sulfide-producing enzyme that enhances drought tolerance in Arabidopsis [J].Plant and Soil,2026,518(1):389-402. |
| [24] | KABIL O, VITVITSKY V, XIE P,et al.The quantitative significance of the transsulfuration enzymes for H2S production in murine tissues[J].Antioxidants & Redox Signaling,2011,15(2):363-372. |
| [25] | KIMURA H.Production and physiological effects of hydrogen sulfide[J].Antioxidants & Redox Signaling,2014,20(5):783-793. |
| [26] | PEDRE B, DICK T P.3-Mercaptopyruvate sulfurtransferase:an enzyme at the crossroads of sulfane sulfur trafficking[J].Biological Chemistry,2021,402(3):223-237. |
| [27] | FRÄSDORF B, RADON C, LEIMKÜHLER S.Characterization and interaction studies of two isoforms of the dual localized 3-mercaptopyruvate sulfurtransferase TUM1 from humans[J].Journal of Biological Chemistry,2014,289(50):34543-34556. |
| [28] | PAPENBROCK J, RIEMENSCHNEIDER A, KAMP A,et al.Characterization of cysteine-degrading and H2S-releasing enzymes of higher plants:from the field to the test tube and back[J].Plant Biology,2007,9(5):582-588. |
| [29] | ALVAREZ C, CALO L, ROMERO L C,et al.An O-acetylserine(thiol)lyase homolog with L-cysteine desulfhydrase activity regulates cysteine homeostasis in Arabidopsis [J].Plant Physiology,2010,152(2):656-669. |
| [30] | WIRTZ M, DROUX M, HELL R. O-acetylserine (thiol) lyase:an enigmatic enzyme of plant cysteine biosynthesis revisited in Arabidopsis thaliana [J].Journal of Experimental Botany,2004,55(404):1785-1798. |
| [31] | LIU Z Q, QU J J, ZHANG L P,et al.Cloning of cucumber LCD and DES gene and their response to abiotic stress[J].Scientia Horticultura,2021,278:109802. |
| [32] | JIN Z P, SUN L M, YANG G D,et al.Hydrogen sulfide regulates energy production to delay leaf senescence induced by drought stress in Arabidopsis [J].Frontiers in Plant Science,2018,9:1722. |
| [33] | LIU D M, LI J, LU J J,et al.Cloning and functional analysis of four O-acetylserine (thiol) lyase family genes from foxtail millet[J].Plant Physiology and Biochemistry,2019,139:325-332. |
| [34] | RIEMENSCHNEIDER A, WEGELE R, SCHMIDT A,et al.Isolation and characterization of a D-cysteine desulfhydrase protein from Arabidopsis thaliana [J].FEBS Journal,2005,272(5):1291-1304. |
| [1] | Lipeng ZHANG, Mei WU, Hongpeng WANG, Tianyu LI. Salt Stress-related RcNAC22 Gene Cloning and Function Analysis in Rhodiolacrenulata [J]. Bulletin of Botanical Research, 2026, 46(2): 246-258. |
| [2] | Zheng LI, Fengxin CHEN, Yuqi LIU, Mingming LI, Jiacan YIN, Chao LIU, Xinli XIA. Overexpression of the PtrMYB002 gene Inhibits Growth of Arabidopsis thaliana and Enhances Its Drought Resistance [J]. Bulletin of Botanical Research, 2026, 46(1): 67-82. |
| [3] | Xiuying MA, Jinke LI, Xiaoyang ZHOU, Shaoliang CHEN. Research Progress of Ca2+-ATPase Involved in Regulation of Plant Salt Tolerance [J]. Bulletin of Botanical Research, 2024, 44(5): 641-654. |
| [4] | Hengfeng ZHANG, Yangwu HE, Huanchao ZHANG, Qingcui WEI. Metabolomics Analysis of Lagerstroemia indica in Response to Salt and Alkali Stress [J]. Bulletin of Botanical Research, 2024, 44(3): 420-430. |
| [5] | Xuefeng HAO, Xiaoyu JIA, Haiyan CAO, Chunxia KANG, Yanxi PEI. Isolation and Identification of a Novel Enlarged Leaf Mutant atscamp in Arabidopsis thaliana [J]. Bulletin of Botanical Research, 2024, 44(2): 232-238. |
| [6] | Wei LIU, Ziqiang ZHU. Recent Advances on Plant Root Thermomorphogenesis [J]. Bulletin of Botanical Research, 2024, 44(1): 1-7. |
| [7] | Fazhi FANG, Huiying GUI, Zhaojia LI, Xiaofeng ZHANG. Physiological Adaptation of Six Mangrove Seedlings to Different Salinity [J]. Bulletin of Botanical Research, 2023, 43(6): 881-889. |
| [8] | Zhuanzhuan JIANG, Li GONG, Yaling SONG. The Chloroplast Division Protein PARC6 Affected the Growth of Cotyledon and Leaf in Arabidopsis thaliana [J]. Bulletin of Botanical Research, 2023, 43(5): 700-710. |
| [9] | Sheng ZHENG, Haixia GAO, Min SU, Shanghuan LU, Tengguo ZHANG, Guofan WU. Exogenous Sucrose Affected AtKEA1 and AtKEA2 to Regulate Root Growth of Seedling in Arabidopsis thaliana [J]. Bulletin of Botanical Research, 2023, 43(4): 562-571. |
| [10] | Lei XU, Xiao XU, Qinsong LIU. Effects of Exogenous Salicylic Acid on Antioxidant System and Gene Expression of Davidia involucrata Seedlings under Salt Stress [J]. Bulletin of Botanical Research, 2023, 43(4): 572-581. |
| [11] | Yuping QIU, Yichuan WANG, Hongwei GUO. Research Progress on the Regulatory Mechanism of Plant Root Hair Development [J]. Bulletin of Botanical Research, 2023, 43(3): 321-332. |
| [12] | Yuanyuan CAI, Jibenben XIA, Wenhan YING, Jieyao WANG, Tao XIE, Kongya XING, Xuanjun FENG, Xuejun HUA. Detailed Phenotypical Analysis on the Mutant ssr1-2 Encoding a Mitochondrial Protein of Arabidopsis thaliana [J]. Bulletin of Botanical Research, 2023, 43(3): 421-431. |
| [13] | Li LI, Xin WANG, Yuejing ZHANG, Lingyun JIA, Hailong PANG, Hanqing FENG. Effects of Abiotic Stresses on the Intracellular and Extracellular ATP Levels of Tobacco Suspension Cells [J]. Bulletin of Botanical Research, 2023, 43(2): 179-185. |
| [14] | Shixian LIAO, Yuting WANG, Liben DONG, Yongmei GU, Fenglin JIA, Tingbo JIANG, Boru ZHOU. Function Analysis of the Transcription Factor PsnbZIP1 of Populus simonii×P. nigra in Response to Salt Stress [J]. Bulletin of Botanical Research, 2023, 43(2): 288-299. |
| [15] | Senyao LIU, Fenglin JIA, Qing GUO, Gaofeng FAN, Boru ZHOU, Tingbo JIANG. Response Analysis of Transcription Factor PsnbHLH162 Gene in Populus simonii × P. nigra under Salt Stress and Low Temperature Stress [J]. Bulletin of Botanical Research, 2023, 43(2): 300-310. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||