Bulletin of Botanical Research ›› 2023, Vol. 43 ›› Issue (4): 622-630.doi: 10.7525/j.issn.1673-5102.2023.04.015
• Molecular biology • Previous Articles Next Articles
Mingyue YUAN1,2, Tianzhong ZHOU2,3, Ma YU1, Bin HU2, Xiangyu LONG2, Hua CHEN1( )
)
Received:2023-03-19
Online:2023-07-20
Published:2023-07-03
Contact:
Hua CHEN
E-mail:hchen@swust.edu.cn
About author:YUAN Mingyue(1998—),female,master student,molecular biology of rubber production from rubber trees.
Supported by:CLC Number:
Mingyue YUAN, Tianzhong ZHOU, Ma YU, Bin HU, Xiangyu LONG, Hua CHEN. Cloning and Functional Analysis of Deubiquitinating Enzyme Gene UCHs from Hevea brasiliensis[J]. Bulletin of Botanical Research, 2023, 43(4): 622-630.
Add to citation manager EndNote|Ris|BibTeX
URL: https://bbr.nefu.edu.cn/EN/10.7525/j.issn.1673-5102.2023.04.015
Table 1
Primer sequences of HbUCH-L3 and HbUCH-L5
引物名称 Primer name | 引物序列(5′→3′) Primer sequence(5′→3′) | 备注 Note |
|---|---|---|
| HbUCH-L3-F | ATTGTGTAGTTCCTTTGGGGCTTAG | HbUCH-L3基因克隆全长引物 Cloning of full-length primers of HbUCH-L3 gene |
| HbUCH-L3-R | CAGAAAATTATTCCCTGCATGTCT | |
| HbUCH-L5-F | AATCCTGAAGATCCAAGCATTTTCC | HbUCH-L5基因克隆全长引物 Cloning of full-length primers of HbUCH-L5 gene |
| HbUCH-L5-R | TGCCAAAAATATGCTAAAAAAGTC | |
| HbUCH-L3-qp-F | TGGAGCGTGCTGCATATCTT | HbUCH-L3实时定量PCR引物 HbUCH-L3 real-time quantitative PCR primer |
| HbUCH-L3-qp-R | AGTTCTCCTTCCACACACGC | |
| HbUCH-L5-qp-F | TGGACTGGTTGCGTATGGTG | HbUCH-L5实时定量PCR引物 HbUCH-L5 real-time quantitative PCR primer |
| HbUCH-L5-qp-R | CCTCCTTTGCAGCTCCTTGA | |
| HbYLS8-qp-F | CCTCGTCGTCATCCGATTC | 实时定量PCR内参引物 Real-time quantitative PCR internal reference primers |
| HbYLS8-qp-R | CAGGCACCTCAGTGATGTC | |
| HbUCH-L3-MBP-F | TTGCGGCCGCATGGTTCCGAAGCCTGTTCTT | HbUCH-L3原核表达引物 HbUCH-L3 prokaryotic expression primer |
| HbUCH-L3-MBP-R | CGGTCGACTTAAATTGCACCTTCAGCTTTC | |
| HbUCH-L5-MBP-F | TTGCGGCCGCATGTCTTGGTGCACTATTGAGT | HbUCH-L5原核表达引物 HbUCH-L5 prokaryotic expression primer |
| HbUCH-L5-MBP-R | CGGTCGACTCATGTTTTTTGCTTGGCCTTC |

Fig.4
Prokaryotic expression analysis of HbUCH-L3(A) and HbUCH-L5(B) proteinA:M.Protein molecular weight marker;1.HbUCH-L3(not purified after induction);2.HbUCH-L3(purification after the first time);3.HbUCH-L3 (purification after the second time);4.HbUCH-L3(purification after the third time);5.HbUCH-L3(purification after the fourth time);B:M.Protein standard molecular weight;1.HbUCH-L5(not purified after induction);2.HbUCH-L5(purification after the first time);3.HbUCH-L5 purification after the second time);4.HbUCH-L5(purification after the third time);5.HbUCH-L5(purification after the fourth time);6.HbUCH-L5(purification after the fifth time)

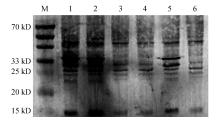
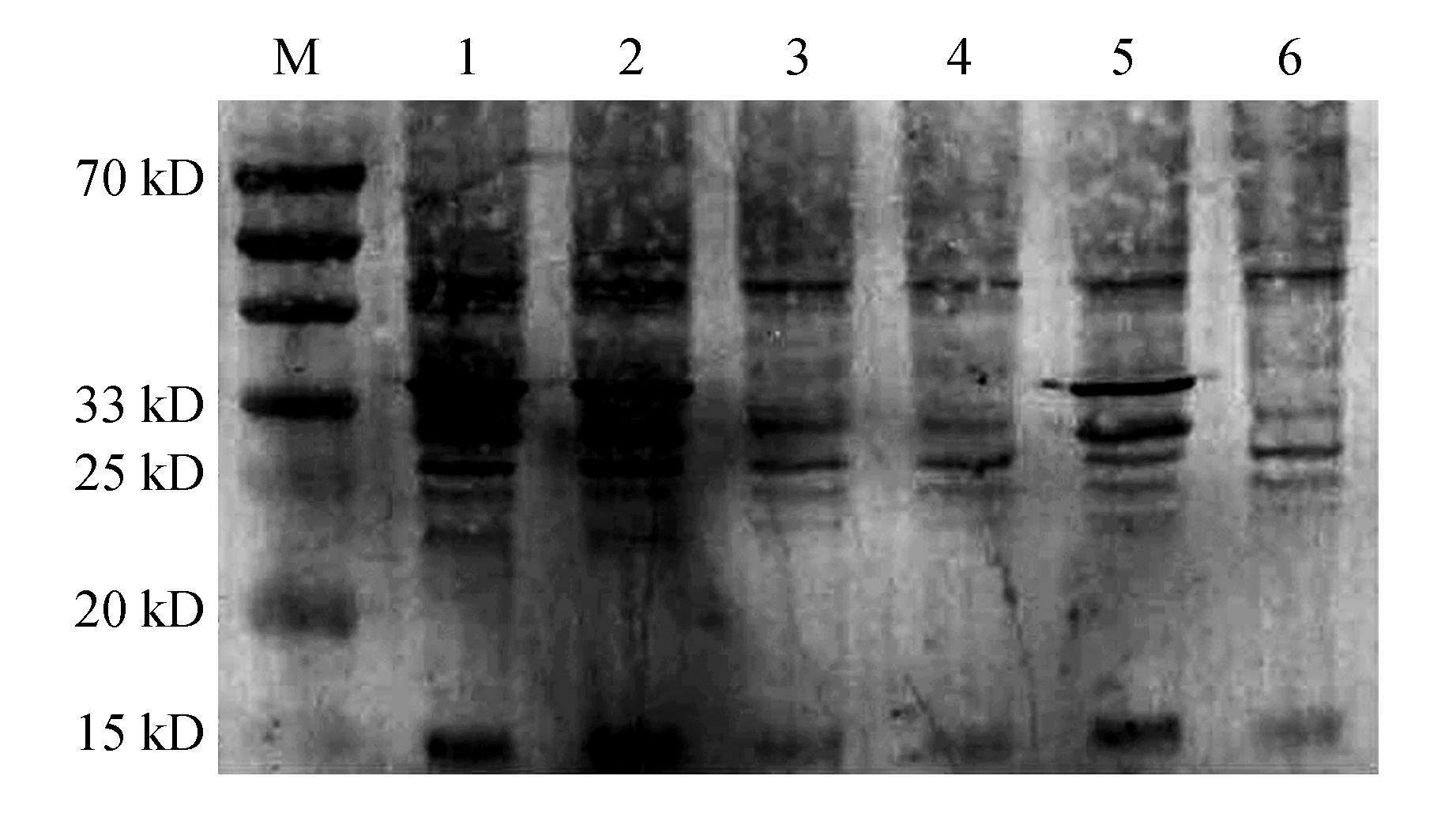
Fig.6
The deubiquitination activity of HbUCH-L3 and HbUCH-L5 protein in C-serum by Western blotM.Protein molecular weight marker;1. 100 μL water and 100 μL C-serum were incubated together;2. 100 μL pMALc5e empty protein wasco-incubated with 100 μL C-serum;3. 50 μL HbUCH-L3 protein was co-incubated with 100 μL C-serum;4. 100 μL HbUCH-L3 protein was co-incubated with 100 μL C-serum;5. 50 μL HbUCH-L5 protein was co-incubated with 100 μL C-serum;6. 100 μL HbUCH-L5 protein was co-incubated with 100 μL C-serum
| 1 | 金华斌,田维敏,史敏晶.我国天然橡胶产业发展概况及现状分析[J].热带农业科学,2017,37(5):98-104. |
| JIN H B, TIAN W M, SHI M J.Current situation and industrial development of natural rubber in China[J].Chinese Journal of Tropical Agriculture,2017,37(5):98-104. | |
| 2 | 安锋,林位夫,谢贵水,等.国内外巴西橡胶树替代作物及技术研发现状[J].热带作物学报,2012,33(6):1134-1141. |
| AN F, LIN W F, XIE G S,et al.Development of alternative natural rubber producing crops and techniques[J].Chinese Journal of Tropical Crops,2012,33(6):1134-1141. | |
| 3 | ZHU J H, ZHANG Z L.Ethylene stimulation of latex production in Hevea brasiliensis [J].Plant Signaling & Behavior,2009,4(11):1072-1074. |
| 4 | ZHANG Y.Transcriptional regulation by histone ubiquitination and deubiquitination[J].Genes & Development,2003,17(22):2733-2740. |
| 5 | 崔超,赵阿慧,李芳,等.泛素/26S蛋白酶体途径调节作物种子大小的研究进展[J].中国农业科技导报,2021,23(9):19-29. |
| CUI C, ZHAO A H, LI F,et al.Research progress of ubiquitin/26S proteasome pathway regulating crop seed size[J].Journal of Agricultural Science and Technology,2021,23(9):19-29. | |
| 6 | 曹孝强,于菲菲,谢旗.泛素化修饰调控植物非生物逆境响应[J].中国基础科学,2020,22(1):7-10. |
| CAO X Q, YU F F, XIE Q.Protein ubiquitination-mediated abiotic stress response in plants[J].China Basic Science,2020,22(1):7-10. | |
| 7 | 田爱梅,于晖,曹家树.植物E3泛素连接酶的分类与功能[J].中国细胞生物学学报,2020,42(5):907-915. |
| TIAN A M, YU H, CAO J S.Classification and function of E3 ubiquitin ligase in plants[J].Chinese Journal of Cell Biology,2020,42(5):907-915. | |
| 8 | 李招娣,兰秋艳,李衍常,等.定量蛋白质组学揭示酵母去泛素化酶Ubp14生物学功能[J].生物工程学报,2022,38(10):3901-3913. |
| LI Z D, LAN Q Y, LI Y C,et al.Quantitative proteomics reveal the potential biological functions of the deubiquitinating enzyme Ubp14 in Saccharomyces cerevisiae [J].Chinese Journal of Biotechnology,2022,38(10):3901-3913. | |
| 9 | 程岚,刘春燕,刘琼,等.植物UBP类去泛素化酶功能研究进展[J].植物生理学报,2023,59(3):433-441. |
| CHEN L, LIU C Y, LIU Q,et al.Advances in functional studies of plant UBP deubiquitinating enzymes[J].Plant Physiology Journal,2023,59(3):433-441. | |
| 10 | KULATHU Y, KOMANDER D.Atypical ubiquitylation:the unexplored world of polyubiquitin beyond Lys48 and Lys63 linkages[J].Nature Reviews Molecular Cell Biology,2012,13(8):508-523. |
| 11 | NIJMAN S M B, LUNA-VARGAS M P A, VELDS A,et al.A genomic and functional inventory of deubiquitinating enzymes[J].Cell,2005,123(5):773-786. |
| 12 | HUANG T T, NIJMAN S M B, MIRCHANDANI K D,et al.Regulation of monoubiquitinated PCNA by DUB autocleavage[J].Nature Cell Biology,2006,8(4):341-347. |
| 13 | DOELLING J H, YAN N, KUREPA J,et al.The ubiquitin‐specific protease UBP14 is essential for early embryo development in Arabidopsis thaliana [J].The Plant Journal,2001,27(5):393-405. |
| 14 | YANG P Z, SMALLE J, LEE S,et al.Ubiquitin C‐terminal hydrolases 1 and 2 affect shoot architecture in Arabidopsis[J].The Plant Journal,2007,51(3):441-457. |
| 15 | LONG X Y, FANG Y J, QIN Y X,et al.Latex-specific transcriptome analysis reveals mechanisms for latex metabolism and natural rubber biosynthesis in laticifers of Hevea brasiliensis[J].Industrial Crops and Products,2021,171:113835. |
| 16 | APPEL R D, BAIROCH A, HOCHSTRASSER D F.A new generation of information retrieval tools for biologists:the example of the ExPASy WWW server[J].Trends in Biochemical Sciences,1994,19(6):258-260. |
| 17 | TANZ S K, CASTLEDEN I, SMALL I D,et al.Fluorescent protein tagging as a tool to define the subcellular distribution of proteins in plants[J].Frontiers in Plant Science,2013,4:214. |
| 18 | ALMAGRO ARMENTEROS J J, TSIRIGOS K D, SØNDERBY C K,et al.SignalP 5.0 improves signal peptide predictions using deep neural networks[J].Nature Biotechnology,2019,37(4):420-423. |
| 19 | KROGH A, LARSSON B, VON HEIJNE G,et al.Predicting transmembrane protein topology with a hidden Markov model:application to complete genomes[J].Journal of Molecular Biology,2001,305(3):567-580. |
| 20 | KUMAR S, TAMURA K,NEI M.MEGA:molecular evolutionary genetics analysis software for microcomputers[J].Bioinformatics,1994,10(2):189-191. |
| 21 | WANG D H, SONG W, WEI S W,et al.Characterization of the ubiquitin C-terminal hydrolase and ubiquitin-specific protease families in rice(Oryza sativa)[J].Frontiers in Plant Science,2018,9:1636. |
| 22 | RONG C, ZHOU R, WAN S,et al.Ubiquitin carboxyl-terminal hydrolases and human malignancies:the novel prognostic and therapeutic implications for head and neck cancer[J].Frontiers in Oncology,2021,10:592501. |
| 23 | 黄浩.泛素羧基端水解酶UCH家族水解反应动力学研究[D].苏州:苏州大学,2009. |
| HUANG H.Study on the activity and dynamics of ubiquitin C-terminal hydrolase[D].Suzhou:Soochow University,2009. | |
| 24 | LEE Y T C, CHANG C Y, CHEN S Y,et al.Entropic stabilization of a deubiquitinase provides conformational plasticity and slow unfolding kinetics beneficial for functioning on the proteasome[J].Scientific Reports,2017,7(1):1-14. |
| 25 | 任禹静,王丰.泛素羧基末端水解酶L3的研究进展[J].生命科学仪器,2020,18(2):14-22. |
| REN Y J, WANG F.Advances in the study of ubiquitin C-terminal hydrolase L3[J].Life Science Instruments,2020,18(2):14-22. | |
| 26 | 牛彤旭,王张,宝鲁日,等.UCH-L3和HIF-1α在乳腺良恶性病变中的表达和临床意义[J].海南医学院学报,2021,27(3):174-179. |
| NIU T X, WANG Z, BAO L R,et al.Expression and clinical significance of UCH-L3 and HIF-1a in breast benign and malignant lesions[J].Journal of Hainan Medical University,2021,27(3):174-179. | |
| 27 | 陈立杰,方远鹏,杜巧丽,等.水稻去泛素化酶OsUCH-L5基因的克隆及表达分析[J].种子,2021,40(3):45-51. |
| CHEN L J, FANG Y P, DU Q L,et al.Cloning and expression analysis of OsUCH-L5 gene encoded rice deubiquitin enzyme[J].Seed,2021,40(3):45-51. | |
| 28 | OLDAK L, CHLUDZINSKA-KASPERUK S, MILEWSKA P,et al.MMP-1,UCH-L1,and 20S proteasome as potential biomarkers supporting the diagnosis of brain glioma[J].Biomolecules,2022,12(10):1477. |
| 29 | 尉娜,路坦,谭军.脑梗死患者静脉溶栓疗程中泛素羧基末端水解酶L1和缺血修饰白蛋白水平的动态变化及临床意义[J].中国心血管杂志,2022,27(3):263-268. |
| WEI N, LU T, TAN J.Dynamic changes and clinical significance of ubiquitin carboxyl-terminal hydrolase L1 and ischemia-modified albumin levels during intravenous thrombolysis in patients with cerebral infarction[J].Chinese Journal of Cardiovascular Medicine,2022,27(3):263-268. | |
| 30 | 丁珊,任禹静,姜凌,等.磷酸化对UCHL3体外去泛素化酶活性的影响[J].生物技术进展,2019,9(5):527-535. |
| DING S, REN Y J, JIANG L,et al.Effect of phosphorylation on deubiquitinase activity of UCHL3 in vitro[J].Current Biotechnology,2019,9(5):527-535. | |
| 31 | WOLBERGER C.Mechanisms for regulating deubiquitinating enzymes[J].Protein Science,2014,23(4):344-353. |
| 32 | PICKART C M, ROSE I A.Ubiquitin carboxyl-terminal hydrolase acts on ubiquitin carboxyl-terminal amides[J].Journal of Biological Chemistry,1985,260(13):7903-7910. |
| 33 | 魏芳,校现周,刘实忠.适合巴西橡胶树胶乳C-乳清蛋白双向电泳技术的建立[J].热带作物学报,2009,30(11):1618-1623. |
| WEI F, XIAO X Z, LIU S Z.Establishment of a two-dimensional electrophoresis technology for latex C-serum of Hevea brasiliensis[J].Chinese Journal of Tropical Crops,2009,30(11):1618-1623. |
| [1] | Bingbing GUO, Mingyang LIU, Longjun DAI, Hong YANG, Lifeng WANG. Research Progress on the Mechanism of Plant Hormone Regulating Yield and Latex Flow in Hevea brasiliensis [J]. Bulletin of Botanical Research, 2024, 44(2): 161-167. |
| [2] | Hong YANG, Lifeng WANG, Longjun DAI, Bingbing GUO. Effects of Tapping Panel Dryness on Mitochondrial Ultrastructure and ROS Metabolism in Barks of Rubber Tree (Hevea brasiliensis) [J]. Bulletin of Botanical Research, 2023, 43(1): 69-75. |
| [3] | Bi QIN, Mingyang LIU, Meng WANG, Lifeng WANG, Fei HUANG. Cloning and Expression Analysis of DELLA gene HbRGL1 from Hevea brasiliensis Müll. Arg [J]. Bulletin of Botanical Research, 2022, 42(6): 997-1004. |
| [4] | Longjun DAI, Mingyang LIU, Jianghua YANG, Kai ZHOU, Bingbing GUO, Hong YANG, Lifeng WANG. Structural and Gene Expression Analysis of a DUF1262 Domain Protein in Latex from Hevea brasiliensis [J]. Bulletin of Botanical Research, 2022, 42(5): 802-810. |
| [5] | Hua-Feng CHEN, Song-Le FAN, Li-Feng WANG. Structural and Functional Analysis of Gibberellin Signaling Transduction Pathway Gene HbRGA1 in Rubber Tree [J]. Bulletin of Botanical Research, 2021, 41(4): 531-539. |
| [6] | WANG Xiang-Jun, ZENG Xian-Zhen, WANG Ya-Jie, HUANG Xiao, ZHANG Yuan-Yuan, LI Wei-Guo. Wood Ultrastructure Observation of Rubber Tree (Hevea brasiliensis Muell.Arg.) Young Stem [J]. Bulletin of Botanical Research, 2018, 38(6): 876-885. |
| [7] | WANG Meng, ZHNG Dong, LU Yan-Xi, XIAO Hua-Xing, ZHENG Fu-Cong, ZHANG Yu. Multiple Roles of Berberine Bridge Enzyme Gene HbBBE1 in Response to Stress in Hevea brasiliensis Muell. Arg. [J]. Bulletin of Botanical Research, 2018, 38(5): 704-713. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||